Carbonic acid.
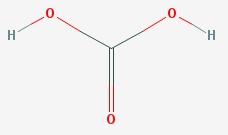
Carbonic acid
Actually, when carbon dioxide is dissolved in water, only a small portion of it reacts with the water to form carbonic acid. Under pressure, more and more of the carbon dioxide reacts, creating more of the acid.
In a soda can or bottle, the pressure is two and a half times as high as normal atmospheric pressure, and the acidity starts to approach that of orange juice. This makes carbonated water taste a little tart (sour, like orange juice).
When the bottle is opened, the pressure drops to normal atmospheric pressure, and the carbonic acid slowly turns back into water and carbon dioxide. This releases the bubbles you see in your drink.
The bubbles in your drink form around nucleation sites. These are places where the solution has a large contact area with a surface, generally and sharp points, edges, scratches, or crevices. If you have a large number of sharp points and crevices, the nucleation proceeds rapidly.
Champagne glasses sometimes have small scratches placed in the bottom to make a nice looking stream of bubbles flow up the glass. The scratches act as nucleation sites. Another now-famous example of nucleation is dropping Mentos candy into a carbonated beverage, and watching the rapid, almost violent release of carbon dioxide from the millions of nucleation sites the candy offers.
